Epidemiology and Changing Patterns of FUO
The epidemiologic profile of FUO has undergone remarkable changes over the past 70 years, reflecting advances in diagnostic technology, changes in antibiotic usage, the emergence of new pathogens, and demographic shifts in the populations at risk for specific causes of fever.
Overall Epidemiologic Trends
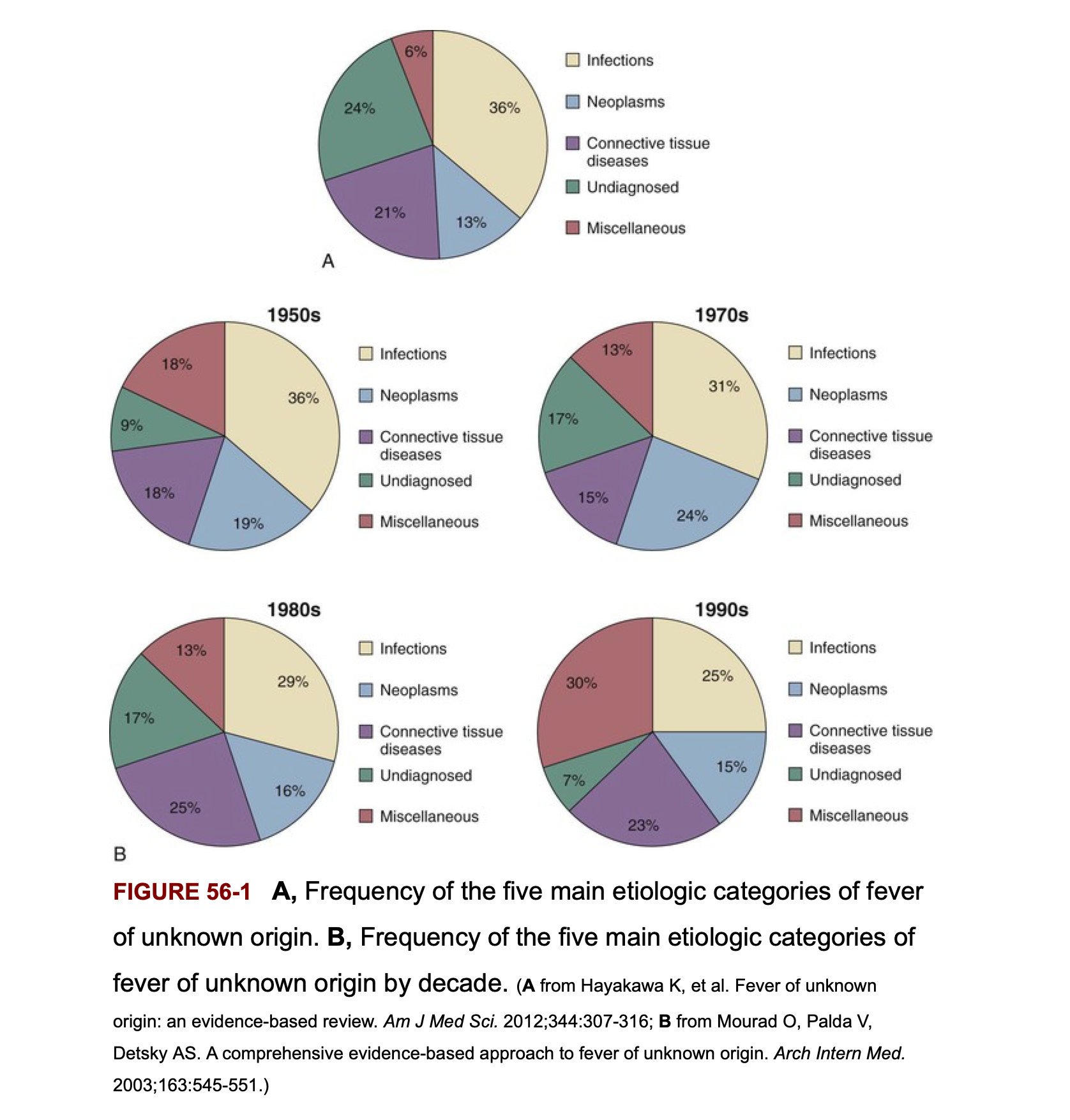
Systematically cataloging the causes of FUO in large patient series reveals that fever of unknown origin can be attributed to one of several broad categories: infections (bacterial, fungal, parasitic, viral), noninfectious inflammatory diseases (NIID), malignancies, miscellaneous causes, and truly undiagnosed cases. The relative proportions of these categories have shifted substantially over time.
In the 1960s and 1970s, infections accounted for approximately 40-50% of FUO cases in developed countries, with tuberculosis, endocarditis, and abscess (both intraabdominal and other sites) being particularly common. Neoplastic causes accounted for 15-20%, connective tissue diseases approximately 15-20%, and miscellaneous causes 10-15%. By the 1990s and 2000s, this distribution had shifted dramatically in developed countries, with infections declining to approximately 25-35% of cases. This decline reflects the impact of more sophisticated diagnostic imaging (CT, MRI, PET), molecular diagnostics, and more effective empiric antibiotic therapy that eliminates some previously undiagnosed infectious causes.
Conversely, noninfectious inflammatory diseases (NIID) have increased as a proportion of FUO cases in developed countries, now accounting for 30-40% of cases. This apparent increase may partly reflect improved recognition of these conditions and greater precision in diagnosis, but it also reflects the genuine decline in infectious causes. Malignancies account for approximately 10-15% of FUO cases and tend to occur more commonly in older patient populations. Miscellaneous causes and undiagnosed FUO account for the remainder.
Geographic Variation
The epidemiologic distribution of FUO causes varies dramatically by geographic region and level of economic development. In low- and middle-income countries, infections remain the leading cause of FUO, accounting for 50-70% of cases depending on the region. Tuberculosis, brucellosis, malaria, and other endemic infections occur at much higher frequency in these settings. In developing countries, environmental factors such as animal exposure, water quality, and prevalence of parasitic and mycobacterial infections all influence the differential diagnosis of FUO.
Timeline of Diagnostic Technology Impact
The introduction and widespread adoption of computed tomography in the 1980s marked a major watershed in FUO diagnosis, dramatically improving the detection of occult abscesses, malignancies, and other structural lesions. The subsequent development of PET-CT imaging in the late 1990s and 2000s further enhanced diagnostic capabilities, particularly for malignancy and certain infections. Molecular diagnostics based on nucleic acid amplification have enabled detection of fastidious organisms previously difficult or impossible to identify. These technological advances have progressively shifted the epidemiologic pattern of FUO, improving diagnostic yield while paradoxically increasing the proportion of cases attributable to NIID (by identifying and excluding infectious causes more effectively).
Classic FUO by Patient Population
The differential diagnosis and clinical approach to FUO varies substantially depending on the patient’s age, comorbidities, and geographic and epidemiologic context. Several patient populations merit specific discussion because their presentations and diagnostic patterns differ markedly from general adult populations.
FUO in Infants and Young Children
In infants under 3 months of age, bacteremia with common neonatal pathogens (group B Streptococcus, Escherichia coli, Listeria monocytogenes) represents the leading concern, and FUO in neonates requires empiric evaluation for bacteremia and meningitis even when signs of localized infection are absent. Bloodstream infections and respiratory infections remain the most common identifiable sources of fever in infants and young children, followed by urinary tract infections.
In children older than 3 months but less than 5 years of age, Kawasaki disease must be considered high in the differential diagnosis, particularly in febrile children presenting with rash, conjunctivitis, and mucosal changes. Failure to recognize and treat Kawasaki disease promptly with intravenous immunoglobulin and aspirin can result in coronary artery complications and lifelong cardiac sequelae, making early recognition essential.
In older children (5-18 years of age), the differential diagnosis broadens to include Epstein-Barr virus infection (accounting for approximately 15% of FUO cases in this age group), osteomyelitis (approximately 10%), systemic-onset juvenile idiopathic arthritis (formerly known as Still disease), bartonellosis (approximately 5%), and urinary tract infections (approximately 4%). Geographic variation is important; for instance, in endemic areas, Q fever and other zoonotic infections account for higher proportions of FUO in children.
FUO in Older Adults
The epidemiologic pattern of FUO shifts significantly in adults above 65-70 years of age, with substantial differences from younger populations. In this age group, noninfectious inflammatory diseases predominate over infections in developed countries. Giant cell arteritis (temporal arteritis) and polymyalgia rheumatica, both autoimmune vasculitic conditions more common in older adults, account for a substantial proportion of FUO cases in this population. Giant cell arteritis can present as fever with only mild systemic symptoms, and the association of fever with headache, jaw claudication, and vision changes should raise immediate suspicion.
When infections are identified in older adults with FUO, they tend to be serious and deep-seated: intraabdominal abscess, complicated pyelonephritis with bacteremia, tuberculosis (both recent infection and reactivation of remote foci), and endocarditis in the setting of underlying valvular disease. Older adults are at increased risk for infections that evade typical diagnostic modalities, such as mycobacterial infections that may not be cultured during standard microbiologic evaluation.
An important distinction between older and younger patients with FUO is that factitious fever is rare in older adults; the vast majority of older patients with documented fever have genuine pathology that will eventually be identified with appropriate investigation. Undiagnosed FUO in older adults generally carries a poor prognosis, as the underlying diagnosis, while not identified during the initial evaluation, is typically eventually discovered to be serious—often a malignancy or deep-seated infection.
FUO in Returned Travelers
Travelers returning from endemic areas with fever present a unique diagnostic challenge and require specific consideration of geographic epidemiology in the differential diagnosis. Malaria stands out as the leading cause of fever in returned travelers, accounting for 27-47.6% of FUO cases in various series of ill returned travelers. The importance of obtaining a travel history and specifically asking about the destinations visited, activities undertaken, and prophylaxis taken cannot be overstated.
Hepatitis A and hepatitis E are common causes of fever in returned travelers, often accompanied by jaundice and elevated transaminase levels that facilitate recognition. However, hepatitis without jaundice can occur, and serologic testing is required for diagnosis. Respiratory tract infections from endemic pathogens are common and may be attributed to common causes until serologic or molecular testing reveals the true etiology.
Dengue fever presents with fever, headache, myalgias, and arthralgia—a combination often referred to as “breakbone fever” due to the severity of pain—and may progress to dengue hemorrhagic fever in a proportion of infected individuals. Typhoid fever, caused by Salmonella Typhi, remains common in areas with poor sanitation and can present as sustained fever often described as “rose spots” rash on the trunk, relative bradycardia, and gastrointestinal manifestations including diarrhea or constipation.
Amebic liver abscess, caused by Entamoeba histolytica, typically presents with fever and right upper quadrant pain and hepatomegaly. This condition is more common in travelers returning from Mexico and Latin America and can be diagnosed by imaging (ultrasound or CT showing a hypodense hepatic lesion) and serology (positive antibodies to E. histolytica).
Acute HIV infection can present with fever in returned travelers, often accompanied by a characteristic maculopapular rash, lymphadenopathy, and mucocutaneous ulceration. Recognition of acute retroviral syndrome is important because antiretroviral therapy initiated during acute infection may offer benefits for immunologic recovery and reduction in viral reservoir.
| Malaria |
27-47.6% |
Periodic fever, hemolysis, thrombocytopenia; blood smear/PCR diagnostic |
| Hepatitis A/E |
10-15% |
Jaundice, transaminitis; serology diagnostic |
| Dengue |
5-12% |
Myalgias, arthralgia, rash; serologic testing |
| Typhoid fever |
5-10% |
Rose spots rash, relative bradycardia, GI symptoms; blood/bone marrow culture |
| Amebic liver abscess |
3-8% |
RUQ pain, hepatomegaly; serology and imaging |
| Acute HIV |
2-5% |
Rash, lymphadenopathy, mucocutaneous ulcers; fourth-generation antigen/antibody test |
| TB |
2-5% |
Insidious onset, night sweats, weight loss; chest X-ray, AFB culture/PCR |
| Q fever |
1-3% |
Severe headache, myalgias; serologic testing |
Postoperative Fever
Fever occurring in the postoperative period deserves specific discussion because it is extremely common yet rarely has an identified infectious source, and it represents a diagnostic challenge for clinicians who must balance the need to identify serious infections with the overuse of empiric antibiotics in noninfectious fever.
Timing and Etiology
The timing of postoperative fever carries important diagnostic implications. Fever occurring within the first 24-48 hours after surgery is almost never infectious; rather, it represents the physiologic inflammatory response to surgical trauma. This early postoperative fever is expected and self-limited, typically resolving over 24-72 hours, and rarely requires intervention beyond supportive care.
Fever developing on postoperative day 3-5 may represent either ongoing inflammatory response to surgery or early infectious complications such as surgical site infection, pneumonia, or urinary tract infection. The location of surgery, the magnitude of tissue trauma, and the patient’s risk factors for infection all influence the likelihood of postoperative infection.
Fever developing after postoperative day 5-7 is much more likely to represent infection, and in this timeframe infectious causes should be actively sought. Surgical site infections typically present with localized signs (wound erythema, drainage, fluctuance) in addition to fever; when these signs are absent, other sources (pneumonia, urinary tract infection, thrombophlebitis) should be sought.
Diagnostic Approach
A careful physical examination is essential, paying particular attention to the surgical wound (for erythema, drainage, dehiscence, or fluctuance), lungs (for signs of pneumonia), urinary system (for evidence of urinary tract infection), and extremities (for signs of thrombosis or thrombophlebitis). When signs of infection are absent or minimal, fever in the first few postoperative days is usually physiologic, and empiric antibiotics are not indicated.
Laboratory evaluation should be tailored to the clinical presentation. When localized signs of infection are present, appropriate culture (wound culture, sputum culture, urine culture) should be obtained. Blood cultures are appropriate when the patient shows signs of systemic toxicity or sepsis. Broad-spectrum empiric antibiotics should not be initiated unless there is objective evidence of infection or clinical deterioration suggesting serious infection.
Fever in Neutropenic Patients
Fever in the setting of neutropenia (absolute neutrophil count below 500 cells/mm³) represents a medical emergency and warrants rapid evaluation and initiation of empiric broad-spectrum antimicrobial therapy, as neutropenic patients are at extreme risk for rapid progression of bacteremia to sepsis and septic shock.
Definitions and Context
Neutropenic fever is defined as a single temperature measurement ≥38.3°C (101°F) or sustained temperature ≥38.0°C (100.4°F) for ≥1 hour in a patient with absolute neutrophil count below 500 cells/mm³. This definition is less stringent than the FUO definition because neutropenic patients are at such high risk for serious infection that any fever warrants empiric evaluation and therapy.
Febrile neutropenia occurs in 10-50% of patients receiving chemotherapy for solid tumors and in approximately 80% of patients with hematologic malignancies receiving intensive chemotherapy. The risk of febrile neutropenia varies with the specific chemotherapy regimen, the duration and severity of neutropenia, and patient risk factors such as advanced age or the presence of significant comorbidities.
Pathophysiology and Clinical Manifestations
The absence of normal neutrophil-mediated inflammatory responses in the neutropenic patient leads to profoundly altered presentations of infection. The classic signs of infection—purulent drainage, warmth, erythema, abscess formation—depend on an adequate neutrophil infiltrate; in the severely neutropenic patient, these signs may be entirely absent despite serious underlying infection. A patient with bacterial pneumonia and an ANC of 100 cells/mm³ may have minimal or no findings on physical examination or even on chest X-ray, yet have life-threatening bacteremia.
For this reason, fever is the only reliable sign of infection in the neutropenic patient, and empiric evaluation and therapy are indicated for any documented fever regardless of physical examination findings. Blood cultures should be drawn from both peripheral blood and from any central venous catheter (if present), as catheter-related bacteremia is a major source of fever in this population.
Table: Clinical Manifestations by Absolute Neutrophil Count
| 500-1000 |
Low |
5-7 days |
May be present |
| 100-500 |
Moderate |
3-5 days |
Often absent |
| <100 |
High |
1-3 days |
Usually absent; may have minimal signs |
Bone marrow examination may be helpful in identifying disseminated fungal infections or mycobacterial infections, particularly when other diagnostic methods have been unrevealing. Cultures of marrow aspirate may grow Mycobacterium avium intracellulare (MAC), Histoplasma capsulatum, or other organisms not readily isolated from peripheral blood.
Empiric Therapy
Empiric broad-spectrum antibiotic therapy is indicated immediately upon documentation of fever in the neutropenic patient, without waiting for culture results. Typical regimens include combination therapy with an anti-pseudomonal beta-lactam (such as piperacillin-tazobactam or a carbapenem) together with either an aminoglycoside or a fluoroquinolone. The choice of regimen depends on local resistance patterns and the patient’s prior antibiotic exposures.
Antifungal therapy (with fluconazole or an echinocandin) is typically added if fever persists for more than 4-7 days despite appropriate antibacterial therapy and neutrophil count remains low. The high incidence of invasive fungal infections (particularly Candida species and Aspergillus) in persistently neutropenic patients necessitates empiric antifungal coverage when bacteremia and other sources have been excluded or inadequately treated.
Fever in Patients with Advanced HIV Disease
The epidemiology of fever in patients with advanced HIV disease has changed dramatically since the introduction of highly active antiretroviral therapy (HAART) in the 1990s. In the era before HAART, fever of unknown origin was extremely common in patients with CD4 counts below 100 cells/mm³ and was frequently the presenting manifestation of serious opportunistic infections.
Primary HIV Infection
Acute HIV infection presents with a mononucleosis-like syndrome in a proportion of infected individuals, typically 1-2 weeks after transmission. Fever, myalgias, arthralgia, and mucocutaneous ulceration may occur, along with lymphadenopathy. A characteristic exanthem consisting of small macules and pustules on the face and trunk is common. The diagnosis of acute retroviral syndrome requires a high index of suspicion, as the symptoms are nonspecific and often attributed to viral gastroenteritis or influenza. Fourth-generation antigen/antibody tests that detect both p24 antigen and antibodies to HIV should be used for diagnosis, as antibody-only tests may be falsely negative during the very early phase of acute infection when antibodies are still developing.
Opportunistic Infections in Advanced HIV
In patients with CD4 counts below 200 cells/mm³ who are not receiving HAART or who have not yet achieved immune reconstitution, opportunistic infections account for the vast majority of febrile episodes. Mycobacterium avium intracellulare (MAC) typically develops in patients with CD4 counts below 50 cells/mm³ and presents as fever, constitutional symptoms, and often disseminated disease with bacteremia. Diagnosis is established by blood culture, which is more sensitive than bone marrow culture for MAC.
Tuberculosis can occur at any CD4 count but is more common in patients with counts below 200 cells/mm³. Both pulmonary and extrapulmonary tuberculosis occur in HIV-positive patients; the presentation may be atypical with minimal findings on chest X-ray in patients with very low CD4 counts.
Cytomegalovirus (CMV) can cause fever in advanced HIV disease, though it typically presents with organ-specific manifestations (retinitis, colitis, esophagitis) rather than primarily as FUO. When CMV presents as fever without organ-specific involvement, diagnosis is challenging and may require blood culture or PCR testing for CMV.
Lymphomas, particularly non-Hodgkin’s lymphoma with a predilection for extranodal sites and central nervous system involvement, occur at increased frequency in HIV-positive patients even in the HAART era. B-cell lymphomas can present with fever and, despite being malignancies rather than infections, may be included in the differential diagnosis of FUO in HIV-positive patients.
Impact of Antiretroviral Therapy
The widespread adoption of HAART has dramatically changed the epidemiology of fever in HIV-positive patients. Patients who achieve adequate immune reconstitution (CD4 count >200 cells/mm³ sustained on therapy) show dramatic reductions in the frequency of opportunistic infections and FUO. However, some patients initially present with advanced HIV disease and very low CD4 counts; in these patients, opportunistic infections may become apparent or worsen shortly after initiation of HAART, a phenomenon called immune reconstitution inflammatory syndrome (IRIS), which can present as fever and may require careful diagnostic evaluation to distinguish from treatment failure or adverse drug effects.
Diagnostic Approach to FUO
The successful evaluation of a patient with fever of unknown origin requires a systematic approach combining detailed history and physical examination, targeted laboratory investigations, radiographic imaging, and in some cases invasive diagnostic procedures. The approach must be tailored to the specific category of FUO and informed by epidemiologic considerations including the patient’s age, geographic history, and comorbidities.
History and Physical Examination
The initial evaluation of any patient with FUO begins with a comprehensive and detailed history, with particular attention to elements that might suggest specific diagnoses. Travel history must be specifically elicited, asking not only about international travel but also about travel within the United States (where endemic mycotic infections, tick-borne illnesses, and other regional pathogens may be encountered). The history should document all travel destinations, duration of stay in each location, activities undertaken (hiking, camping, swimming, animal exposure), and any prophylactic medications taken.
A thorough occupational history is essential, as certain occupations carry specific disease risks. Healthcare workers have increased risk of tuberculosis and bloodborne pathogen exposure. Construction workers and others with environmental exposures may encounter fungi (such as Histoplasma, Coccidioides, Blastomyces) that cause endemic mycoses. Veterinarians and animal handlers have risk of zoonotic infections including brucellosis, leptospirosis, and Q fever.
- Recent travel, including domestic travel and specific activities
- Occupational exposures (construction, healthcare, animal handling)
- Animal exposure (pets, wildlife, farm animals)
- Medication history (including new medications or recent medication changes)
- Family history (familial fever syndromes)
- Immunization status and recent vaccinations
- Sexual history and practices
- Intravenous drug use
- Food and water exposures
The history should also document contact with ill individuals, both in the home and workplace, and should inquire about the source of drinking water (well water carries risk of infections such as leptospirosis and other water-borne pathogens). Medication history is essential, as certain medications can cause drug fever. A family history should be elicited, as familial periodic fever syndromes (such as familial Mediterranean fever) may present with FUO.
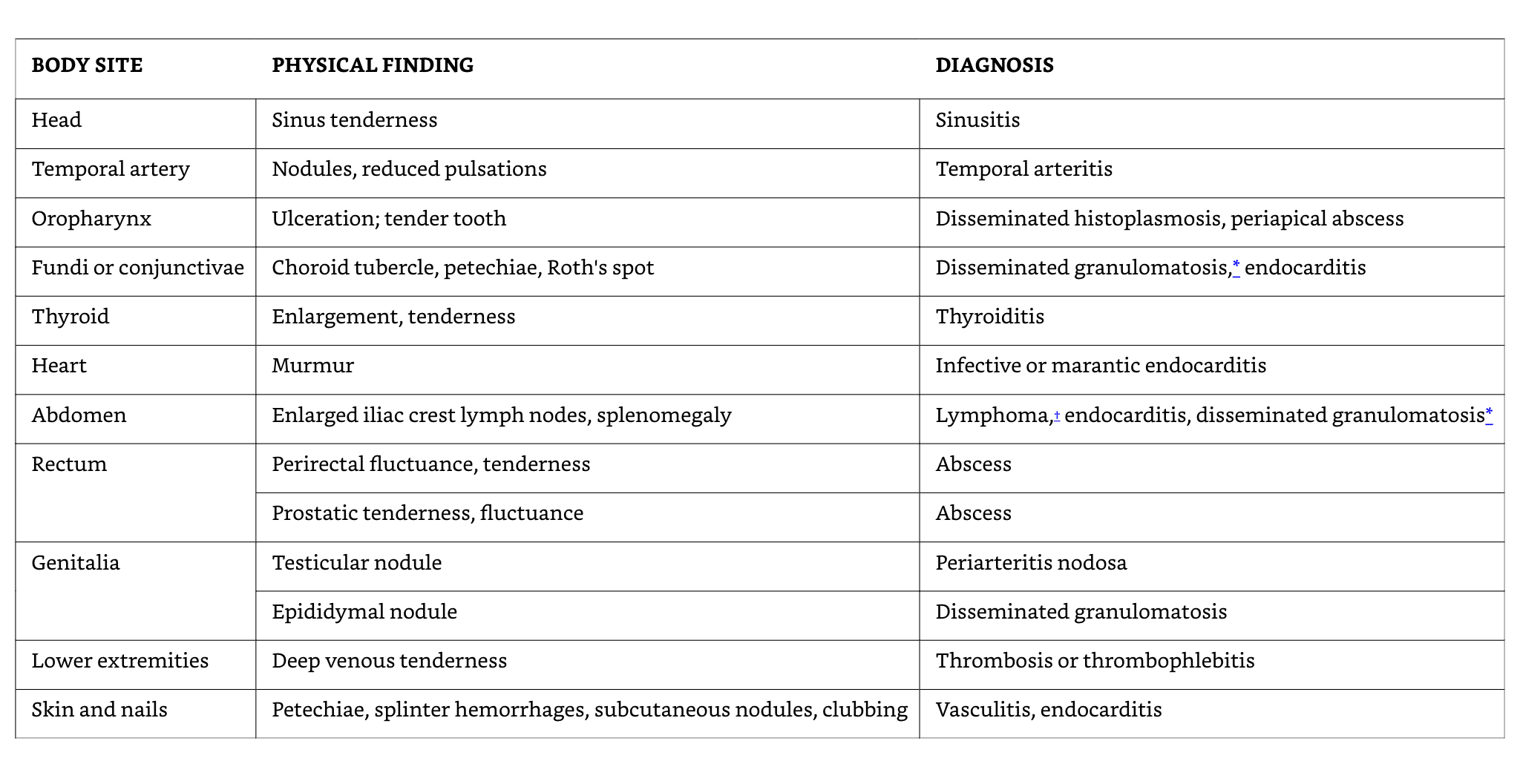
Physical examination must be thorough and repeated; many patients with FUO have subtle physical findings that may not be apparent on initial examination but become evident with repeated careful examination. The skin should be examined closely for rash (which may be evanescent in dengue fever or early typhoid fever), eschars or inoculation sites (suggesting tick-borne diseases or cutaneous anthrax), and vasculitic lesions. The oral cavity, including the teeth and gums, should be examined (as endocarditis may present with Osler nodes on the fingertips, Janeway lesions on the palms and soles, splinter hemorrhages under the nails, and petechiae in the oral vault). The abdomen should be examined for hepatosplenomegaly, masses, and areas of tenderness, as occult malignancy or infection may manifest with organomegaly. The extremities should be examined for signs of thrombophlebitis and for nodes in the epitrochlear and inguinal areas.
Auscultation of the heart may reveal murmurs suggestive of endocarditis; careful auscultation of the lungs may reveal subtle findings of pneumonia or other pulmonary pathology. The fundus should be examined for Roth spots and the conjunctiva for petechiae (both seen in endocarditis). The lymph nodes, including axillary, inguinal, supraclavicular, and occipital nodes should be specifically examined and any nodes palpated noted for size, consistency, and tenderness.
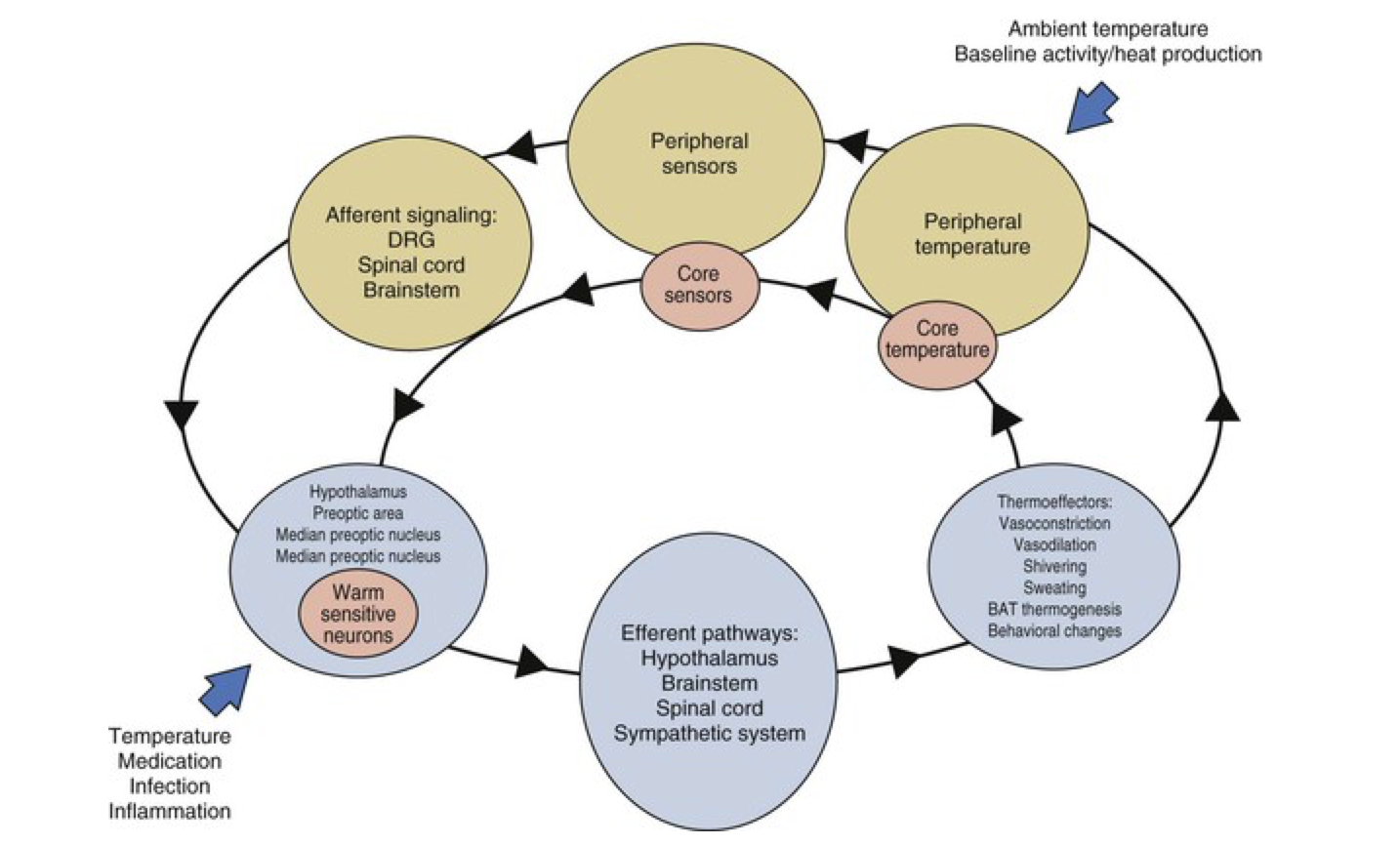
Fever Patterns and Their Diagnostic Significance
While fever patterns have less diagnostic specificity than previously believed, certain patterns remain suggestive of particular diagnoses and merit consideration in the differential diagnosis.
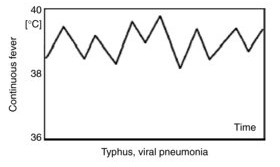
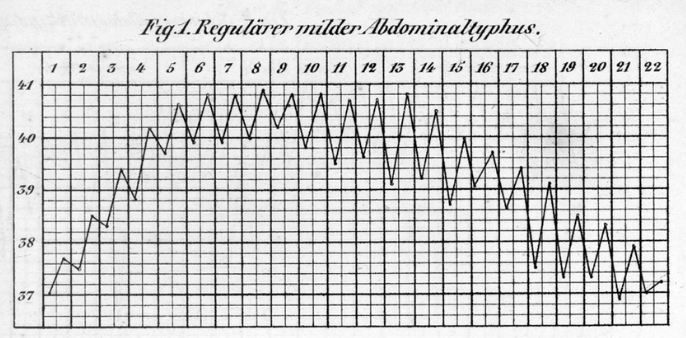
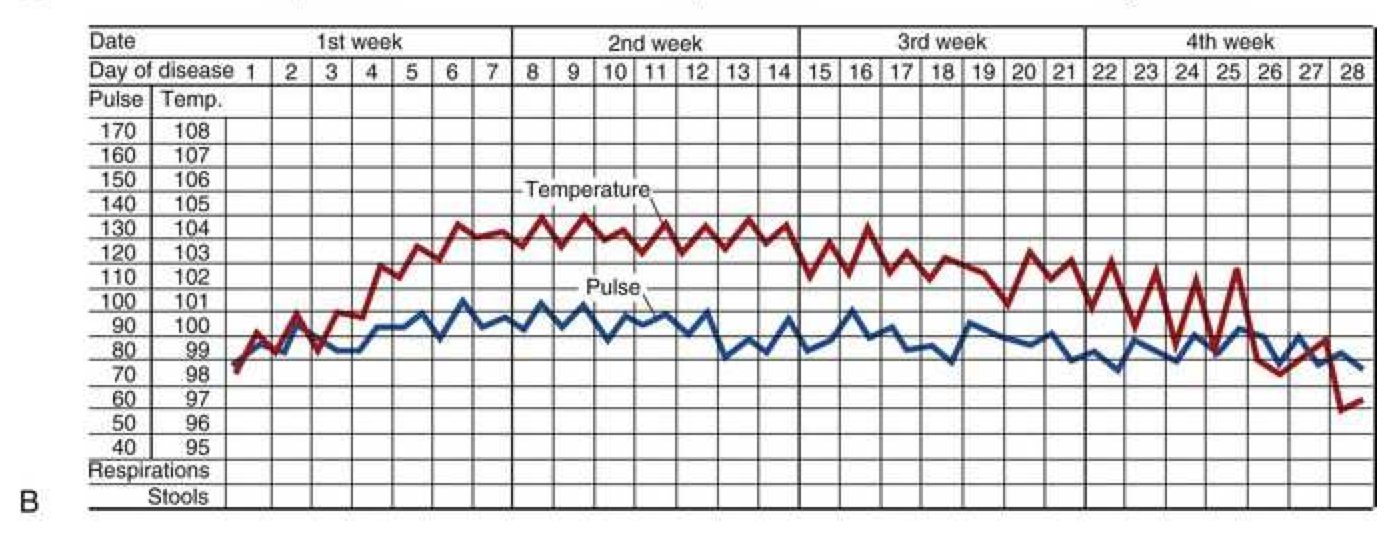
Continuous or sustained fever, in which temperature remains elevated throughout the day without significant fluctuation (varying less than 0.5°C), is typical of pneumonia, rickettsiosis, typhoid fever, CNS infections (meningitis, encephalitis), and falciparum malaria. Patients with sustained fever typically appear quite ill and often have tachycardia, tachypnea, and other signs of significant systemic illness.
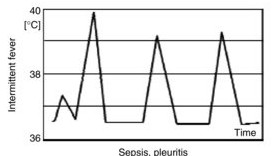
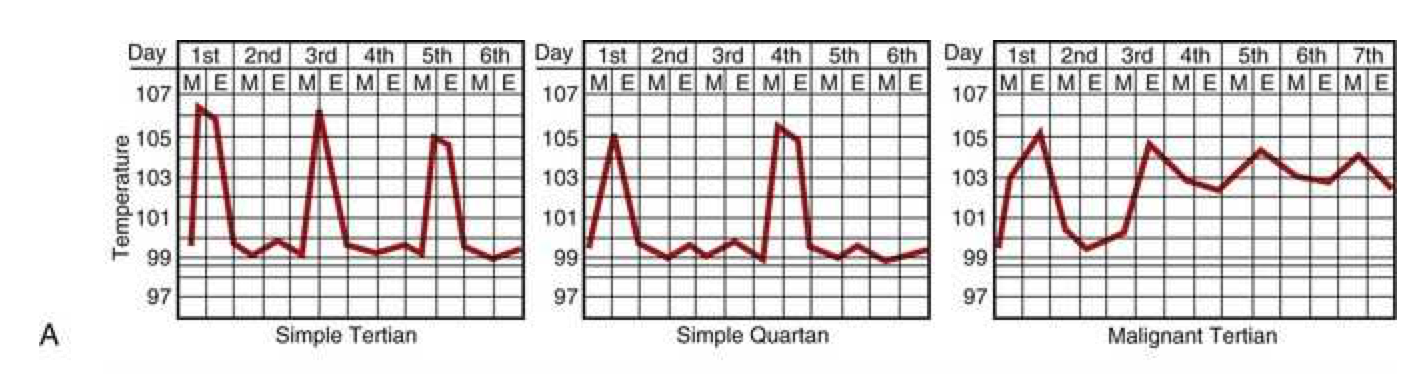
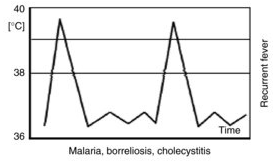
Intermittent or quotidian fever, in which fever spikes occur daily (usually in the late afternoon or evening) with return to normal or near-normal temperature, is characteristic of pyogenic infections such as osteomyelitis, endocarditis, and abscesses. Malaria classically presents with quotidian fever (fever every day), though tertian fever (every other day) and quartan fever (every third day) patterns also occur depending on the Plasmodium species involved.
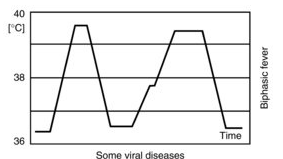
Saddle-back or biphasic fever pattern, characterized by two separate fever spikes separated by a period of relative defervescence, is classic for dengue fever, yellow fever, and Colorado tick fever. The biphasic pattern results from the initial viremic phase, followed by a period of viral control, and then recurrence of viremia.
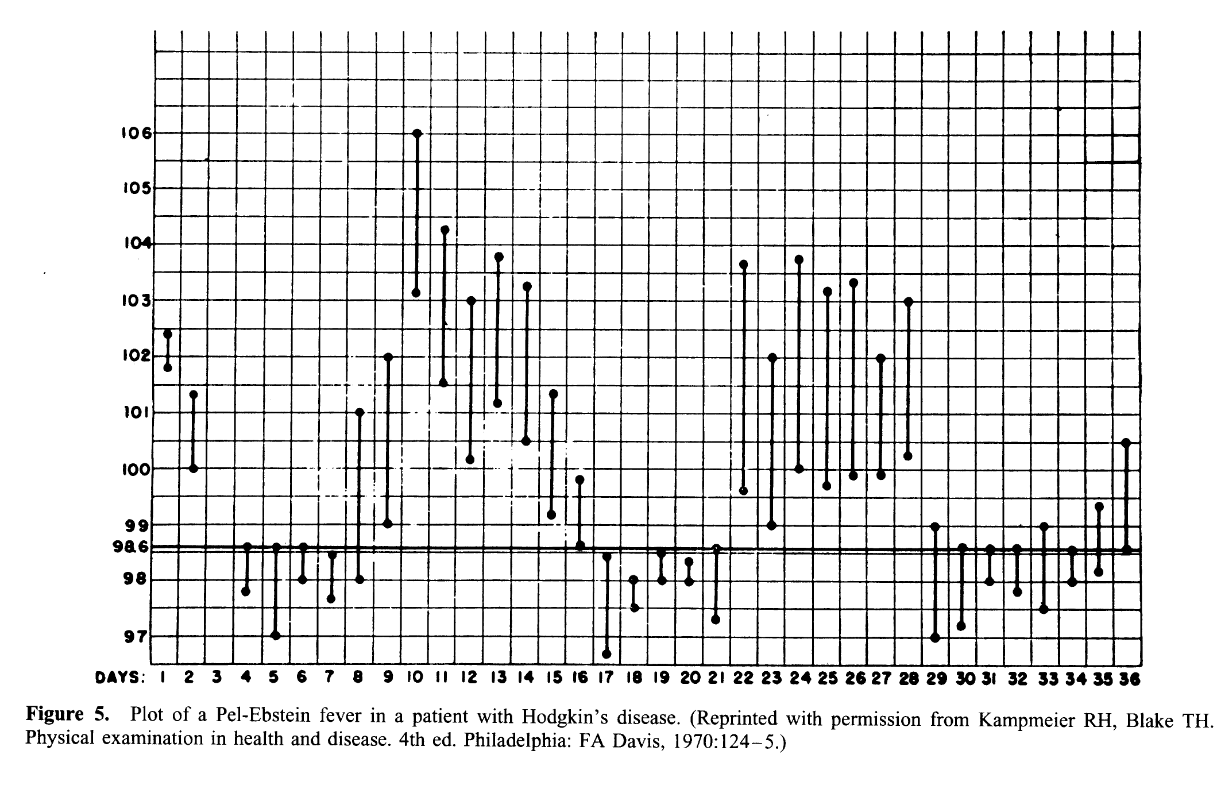
Pel-Ebstein fever, an intermittent pattern with periods of fever alternating with periods of afebrile or hypothermic days, was historically associated with Hodgkin’s disease but is now recognized to occur with other conditions including brucellosis and is not sufficiently specific to be diagnostically useful.
Typus inversus, in which the normal diurnal pattern of temperature variation is inverted (with the nadir in the afternoon and zenith in the early morning), is described in miliary tuberculosis, salmonellosis, and hepatic abscess, though this pattern is not frequently encountered and is not sufficiently specific to be relied upon for diagnosis.
Double quotidian fever, in which there are two fever spikes per day, is described in patients with certain types of endocarditis (particularly gonococcal or meningococcal), miliary tuberculosis, and salmonellosis.
The Jarisch-Herxheimer reaction, a paradoxical exacerbation of fever and systemic symptoms that can occur within hours of initiating therapy for spirochetal infections (particularly syphilis and leptospirosis), reflects rapid release of endotoxin from killed organisms and usually self-resolves within 24-48 hours.
While fever patterns can provide diagnostic clues, they should not be relied upon as the primary diagnostic tool. Many conditions can present with atypical fever patterns, and the same pattern can be seen in diverse conditions. Fever pattern should inform but not direct the diagnostic workup.
Verification of Fever
A surprisingly common finding in the evaluation of patients referred for FUO is that the fever is not actually documented; approximately 30% of referrals for FUO may not have objectively documented fever on careful review. Patients may have subjective sensation of fever, chills, and malaise without actually having elevated body temperature. In some cases, patients may have had fever at the referring institution but have resolved fever by the time they reach the tertiary care center. For this reason, careful documentation of temperature measurements is essential, and in some cases hospitalization for repeated temperature monitoring may be necessary to verify that true fever is present.
Laboratory Investigations
Laboratory evaluation of FUO should be systematic and directed by the clinical presentation rather than consisting of shotgun panels of tests. Initial studies should include complete blood count with differential, comprehensive metabolic panel, and liver function tests. These tests provide important information about the degree of inflammation (elevated white blood cell count, elevated bands), cytopenias (suggesting malignancy or certain infections), renal dysfunction (suggesting infections such as acute glomerulonephritis from endocarditis), and hepatic dysfunction (suggesting infections or malignancies affecting the liver).
Inflammatory markers including erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) are commonly elevated in FUO and help confirm the presence of inflammation, though they are nonspecific. The ESR can remain elevated for weeks to months after resolution of acute illness and is less useful in chronic FUO. CRP tends to normalize more rapidly and may be more useful for monitoring response to therapy.
Blood cultures should be obtained before any antimicrobial therapy in patients with suspected bacteremia or endocarditis. Multiple blood cultures (typically 2-3 sets) increase the likelihood of isolating the pathogen and help distinguish contamination from true bacteremia. Special culture media and extended incubation periods may be needed for fastidious organisms.
Serology should be directed by the clinical and epidemiologic context. Specific serologic tests for infections such as Lyme disease, Q fever, bartonellosis, brucellosis, and leptospirosis should be ordered based on the patient’s travel and exposure history. Serologic testing for autoimmune conditions including antinuclear antibodies (ANA), rheumatoid factor (RF), and markers of vasculitis (antineutrophil cytoplasmic antibodies [ANCA]) should be considered in patients with features suggestive of autoimmune disease.
Blood smears should be examined for microorganisms in patients with travel to malaria-endemic areas; multiple thick and thin smears should be obtained if malaria is suspected, and rapid diagnostic tests (such as rapid antigen detection or PCR) are more sensitive than microscopy. Louse-borne and tick-borne relapsing fevers may be detected on blood smears stained with Wright-Giemsa stain.
Bone marrow examination can be extremely valuable in FUO evaluation in selected cases. A bone marrow aspirate and biopsy should be obtained when granulomatous infection (such as tuberculosis, histoplasmosis, or brucellosis) is suspected, when hematologic malignancy is in the differential diagnosis, or when other diagnostic methods have been unrevealing. The marrow can be cultured for mycobacteria, fungi, and other organisms, and can be examined for granulomas, malignant cells, or other diagnostic features.
Imaging Studies
Computed tomography has become the single most important imaging modality in the evaluation of FUO, with sensitivities ranging from 60-92% depending on the specific site of involvement and the pathology being sought. CT of the abdomen and pelvis is particularly valuable for identifying occult abdominal pathology including occult abscess, necrotic lymph nodes suggesting tuberculosis or lymphoma, splenic infarction, or other sources of fever. CT of the chest is valuable for identifying pneumonia, mediastinal pathology, and other thoracic sources of fever.
Magnetic resonance imaging excels in evaluation of certain conditions, particularly those involving the central nervous system and those affecting the vascular system. MRI is superior to CT for detecting vasculitis affecting the aorta and major vessels; giant cell arteritis shows characteristic vessel wall enhancement on MRI, and MRI is valuable for detecting vasculitis in other conditions including Takayasu’s arteritis and granulomatosis with polyangiitis (GPA, formerly known as Wegener’s granulomatosis). Brain MRI is valuable for detecting encephalitis, meningitis, and other CNS pathology.
¹⁸Fluorodeoxyglucose positron emission tomography combined with CT (¹⁸FDG-PET/CT) has emerged as an extremely valuable tool in FUO evaluation, with reported sensitivities of 86-98% in various series. PET/CT is particularly valuable for detecting occult infections (such as osteomyelitis and deep abscesses), malignancies, and vasculitis. The high sensitivity of PET/CT has led to recommendations that it be performed relatively early in the diagnostic evaluation of FUO rather than being reserved for cases where standard imaging has been unrevealing.
Gallium scans and indium-labeled white blood cell scans, which were historically used for infection and inflammation detection, have largely been replaced by PET/CT due to superior sensitivity and faster acquisition. However, these nuclear medicine studies may still be useful in specific circumstances, particularly in patients where PET/CT is contraindicated or unavailable.
Echocardiography should be performed in any patient with suspected endocarditis, as transthoracic echocardiography can visualize vegetations on valves, and transesophageal echocardiography provides superior resolution and should be performed if transthoracic echocardiography is negative but clinical suspicion for endocarditis remains high. Echocardiography is also valuable for assessing cardiac structure and function in patients with suspected cardiac pathology as a cause of fever (such as myocarditis).
Invasive Diagnostic Procedures
Biopsy of affected tissue may be necessary to establish diagnosis in some cases of FUO. Lymph node biopsy is valuable when lymphadenopathy is present, as histopathology may reveal granulomas suggesting tuberculosis or fungal infection, or may reveal malignancy. Liver biopsy can similarly reveal granulomas or malignancy. Muscle biopsy may be useful in suspected myositis or vasculitis. The yield of biopsy, defined as the proportion of biopsies that result in a diagnostic finding, varies from 30-50% depending on the biopsy site and the clinical presentation; on average, 2-3 separate biopsies may be needed to establish diagnosis.
Laparoscopy and laparotomy have become less frequently used in the diagnostic evaluation of FUO with the advent of superior imaging modalities (CT, PET/CT), but remain valuable tools in selected cases. Laparoscopic evaluation allows direct visualization of intraabdominal structures and collection of tissue for diagnosis; it is less invasive than open laparotomy but provides less extensive visualization. Laparotomy may be justified in cases where a specific abdominal pathology is suspected and other diagnostic methods have been unrevealing, such as when peritoneal carcinomatosis, disseminated abdominal tuberculosis, or small-bowel Crohn disease is suspected.
Molecular and Advanced Diagnostics
Next-generation sequencing (NGS) of microbial nucleic acids is emerging as a powerful diagnostic tool for fastidious organisms that may not grow in culture. 16S rRNA gene sequencing can identify bacterial pathogens from culture-negative specimens or directly from clinical specimens such as blood or tissue. 18S rRNA gene sequencing similarly provides identification of fungal pathogens. These techniques are particularly valuable in cases where conventional culture has been unrevealing.
Molecular genetic testing using panel-based or single-gene approaches can identify monogenic periodic fever syndromes, including familial Mediterranean fever (MEFV gene mutations), tumor necrosis factor (TNF)-receptor-associated periodic syndrome (TNFRSF1A mutations), mevalonate kinase deficiency (MVK mutations), and cryopyrin-associated periodic syndromes (NLRP3 mutations). These rare genetic conditions can present as FUO and should be considered, particularly in younger patients with recurrent episodes of fever.